RECARTA PROCEDURE
The only autologous chondrocyte-based cartilage therapy in Central Eastern Europe
IDENTIFYING THE RIGHT PATIENT
Checking the patient's medical history is crucial for better clinical understanding and prognosis. Do take note of the patient's concomitant medication and co morbidities, if any. Do ensure the patient is currently not associated with any of the following:
On-going chemotherapy/radiation treatment
Previously undergone organ or bone marrow transplant in the last 6 months
Pregnancy, breast-feeding
Patients aged below 18 years
Degenerative joint changes due to osteoarthritis and rheumatoid arthritis
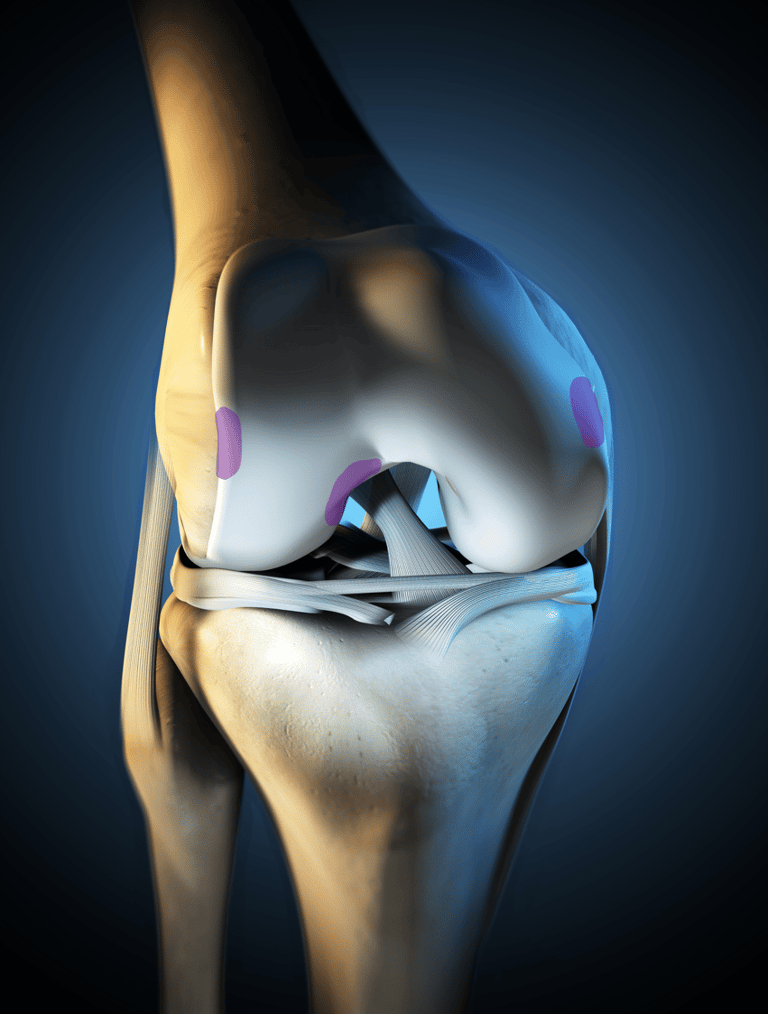
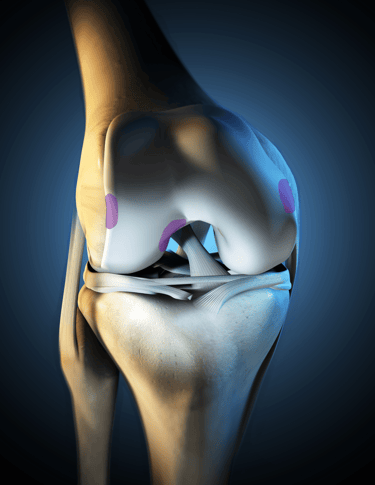
Confirm the Stage of Cartilage Damage: Magnetic Resonance Imaging can be used to determine the Stage and Extent of Cartilage Damage
RECARTA is applicable for ICRS stages (Stage III-IV) without subchondral bone damage
Assessment of candidacy for RECARTA
Staging of knee cartilage damage
Arthroscopic assessment – determining defect size, location, bone involvement
Step 1. CARTILAGE TISSUE BIOPSY
Confirm patients candidacy
Cartilage tissue harvest
A small biopsy of healthy cartilage is taken arthroscopically from a non-weight-baring area of the patient's knee joint (intercondylar notch, the proximal aspect of the medial or lateral femoral condyle) and packaged to RECARTA cartilage tissue harvest transport kit
Coordinate with KELI Therapeutics and the patient to find a suitable RECARTA cell therapy implantation date and time
Refer to the Cartilage Tissue Harvest Manual for the detailed treatment procedure
Step 2. RECARTA PROCESS
The biopsy is shipped to the KELI Therapeutics state-of-the-art facility that provides optimal RECARTA product safety and quality
Chondrocytes are isolated from the biopsy, and cells are cultured for 24±4 days. Rigorous in-process controls and release quality control testing are performed
RECARTA is delivered to a treatment facility at a controlled dose of 1,33 – 2,67 mln. cells per cm2 of defect size


Step 3. RECARTA IMPLANTATION
The cartilage defect is debrided to the healthy, stable cartilage with the ring curette
RECARTA is implanted with a supplied commercial collagen membrane. Use the template provided with the membrane to measure the cartilage defect and cut the membrane to the exact size of the cartilage defect
Before surgery, please Refer to the Implantation manual for step-by-step guidance
RECARTA cells are seeded on the porous side of the membrane and let attach for 10 minutes
RECARTA is implanted in the cartilage defect and checked for stability


